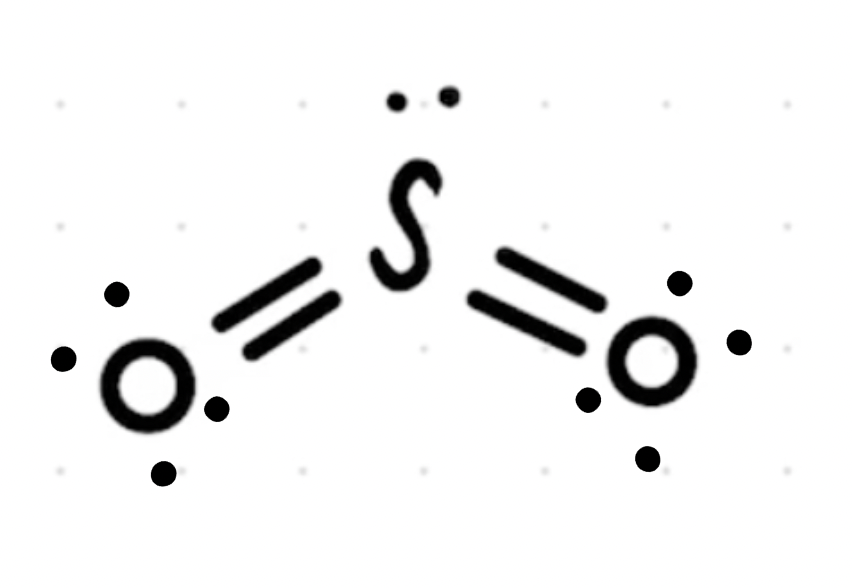
Hey Daniella, The electrons are still there, what you'll notice as we move through Organic Chemistry is that the lone pairs won't always be shown but you will still be expected to know that they are there. It's like the hydrogens on Carbons that we saw aren't shown (this will be explained later on if you have gotten there yet) and it's a quicker way to draw structures.




Replied on Lesson: Resonance and Arrow Pushing
10 Sep 08:29
That would give nitrogen 4 bonds and no formal charge, which violates the octet rule and formal charge rules. If nitrogen is making 4 bonds (2 double bonds), it’s sharing 8 electrons, but nitrogen only has 5 valence electrons. Valid resonance structures share the same total number of electrons, same atom connectivity, and obey the octet rule.
The two resonance forms shown, where the double bond switches between oxygens are the only valid ones.
After you watch this I highly recommend the next two videos they will help you practice these concepts even more:
Study With Us
Practice Problem: Drawing All Resonance Forms That Stabilize a Carbocation