Hi Esteban!
That's a great question! For this particular example, stereochemistry was not shown.
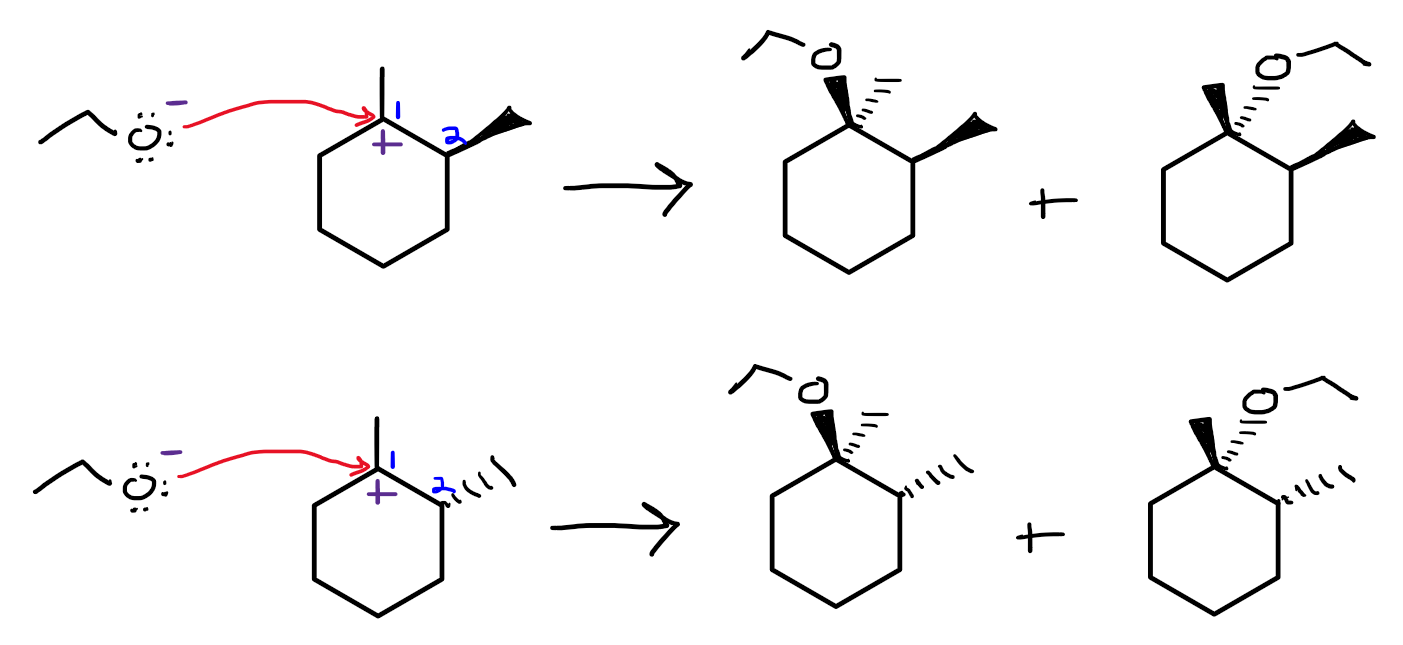
See the attached image. If we consider the stereochemistry, and we define carbon 1 to be the positively charged carbon of the carbocation, a new chiral center is formed at carbon 2 when the methyl shift occurs, giving two enantiomers of the carbocation intermediate--one with the methyl group on carbon 2 wedged, and the other dashed.
The carbocation intermediate is planar, so the nucleophile can attack from either side, giving two enantiomers from each carbocation. So in total, we have four stereoismers.
-Ben


Replied on Lesson: Grignard Reagents and Retrosynthesis
07 Feb 04:41
Hi Riya!
The second reaction of the top row is incorrect. Grignard reagents do not react with alkyl halides via SN2 to form alkanes. If we wanted to prepare propane from an alkyl halide, we would start with propyl bromide, treat it with magnesium to form propylmagnesium bromide, then treat it with a weak acid like water, an alcohol, or a carboxylic acid to protonate it (see image).
Same thing with the second reaction of the middle row. Alkyllithium reagents do not react with alkyl halides to form alkanes. You're right that the alkyllithium would first need to be treated with CuI to form a Gilman reagent before it can react with the alkyl halide.
For the bottom row, not only would the alkyllithium need to be converted to an alkyl halide for the reaction to work, but the alkene shown above the arrow in the second reaction would need to be converted to an alkyl halide in order for the coupling reaction to take place.
-Ben