Hi Pradhikshan,
Although carbon and hydrogen are different elements and have different electronegativities, the difference in electronegativity between carbon and hydrogen is quite small, so in most contexts a C-H bond is considered nonpolar. In this case, these four molecules can be compared by ignoring the polarity of the C-H bond and considering the geometry of the C-Cl bonds.
-Ben


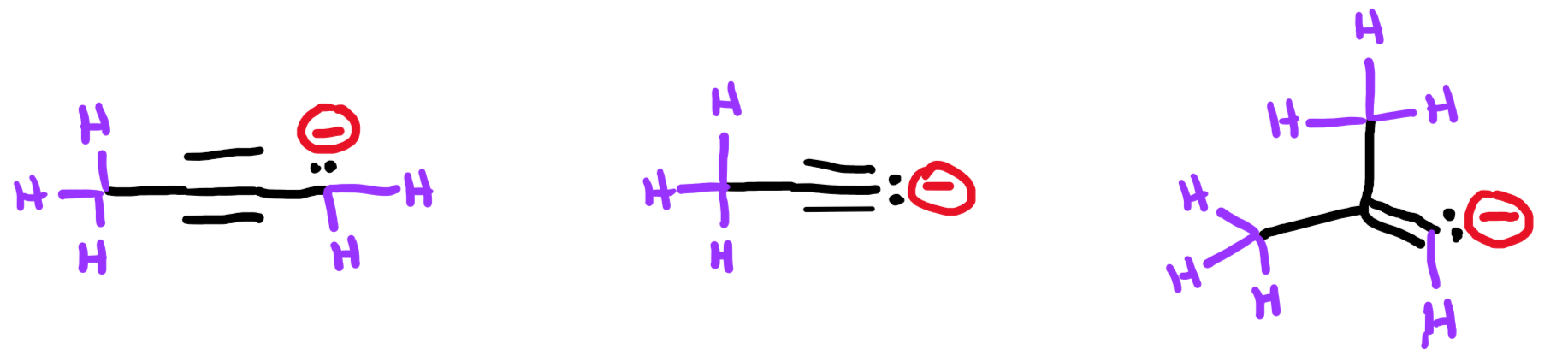
Replied on Lesson: SN2 Reactions
18 Oct 05:42
Hi Mano,
The attached image shows two structures--one in which the negative charge (and the lone pair that attacks the electrophile) are on the carbon atom, and one in which the negative charge is on the nitrogen atom.
If we determine the formal charge of both atoms for both scenarios, we'll see the following:
Structure 1:
• Carbon: 4 valence electrons - (5 electrons in lone pairs/bonds) = -1
• Nitrogen: 5 valence electrons - (5 electrons in lone pairs/bonds) = 0
Structure 2:
• Carbon: 4 valence electrons - (3 electrons in lone pairs/bonds) = +1
• Nitrogen: 5 valence electrons - (5 electrons in lone pairs/bonds) = -2
From the information above, we can see that the first structure, with the nucleophilic center at the carbon atom, has fewer non-zero formal charges, which allows more stability.
If we imagine the nucleophilic attack taking place from the nitrogen atom, we'll see a similar story in the product--we get non-zero formal charges for both the carbon and the nitrogen.
-Ben