For the 22:30 example, yes, you can compare the acidity of different hydrogens within the same molecule by looking at hybridization. In trans-2-butene (the molecule on the right), there are two different types of carbon and two different types of hydrogen. The internal carbons are sp2 hybridized, and the terminal carbons are sp3 hybridized. Since acidity increases with increasing s character, we would predict that the hydrogens bonded to the internal carbons are more acidic than the hydrogens bonded to the terminal carbons.
-Ben


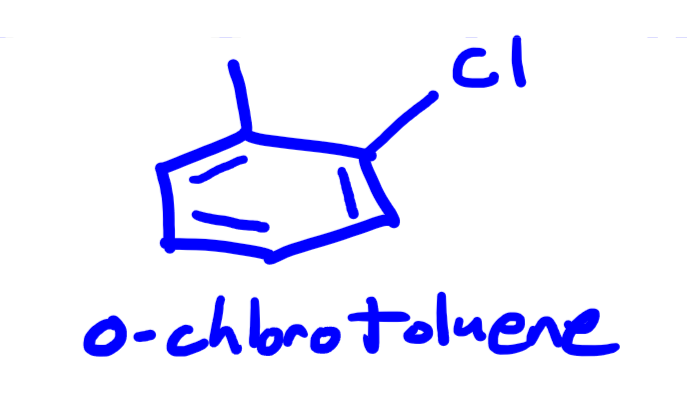
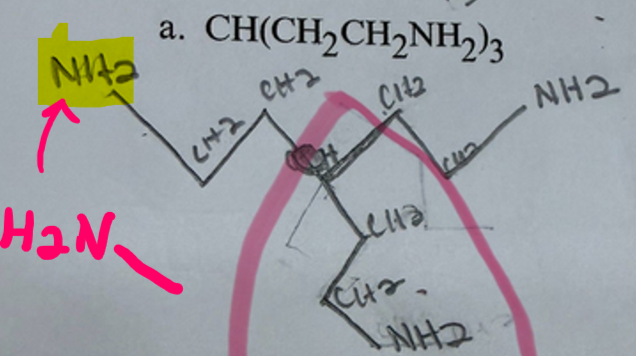
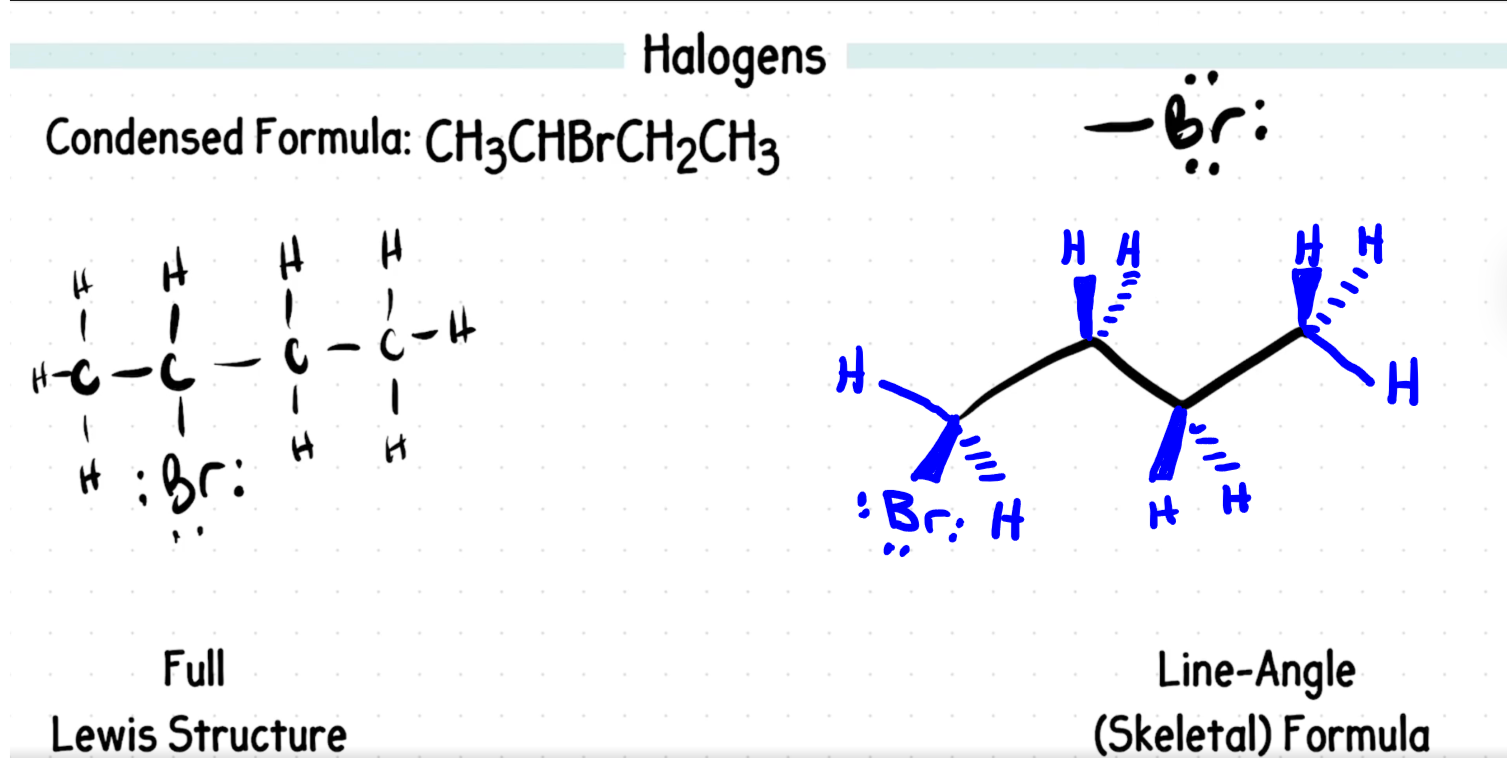
Replied on Practice Problem: Naming Alkenes and Alkene Containing Compounds
16 Sep 04:23
Hi Katheryn,
Yes, I should have specified where the E is located. In this particular molecule, there's only one double bond, so it's unambiguous that the E configuration applies to the double bond between carbons 3 and 4, but if we're strictly following the rules of IUPAC nomenclature, we give E or Z configurations a locant in the name every time no matter what.
Thank you for catching that!
-Ben