Hi Belle,
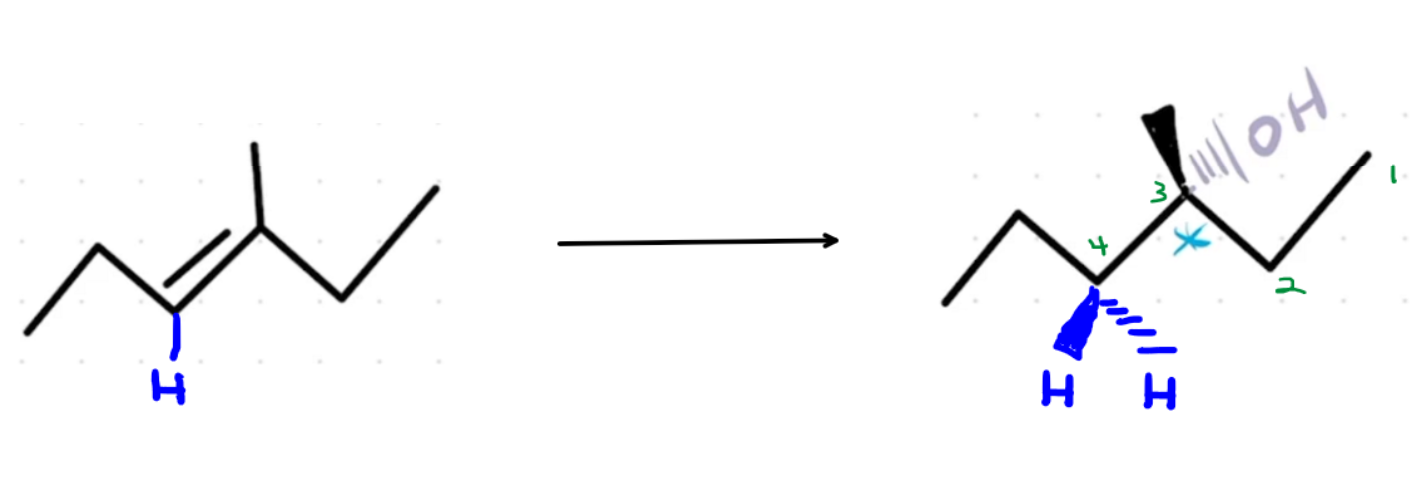
I don't see a halogen anywhere at that timestamp. Do you mean the hydroxyl?
Generally, we use wedged and dashed lines to show the configuration of a chiral center (R or S). If there's no chiral center, then we don't need to use wedged and dashed lines.
Carbon 4, where the hydrogen is added, already had a hydrogen before the reaction. When the product forms, Carbon 4 is bonded to two hydrogens and two carbons. Since it doesn't have four unique groups bonded to it, it's not a chiral center, so we don't need to use wedged and dashed lines to show stereochemistry.



Replied on Lesson: Hydration of Alkenes and Addition of Alcohols
06 Apr 19:24
Hi Belle,
The product of the reaction in the first problem doesn't have any chiral centers. The two carbons that are part of the six-membered ring are equivalent, so there aren't four unique groups bonded to the 3° carbon. Since it doesn't have four unique groups bonded to it, it's not a chiral center, so there's no enantiomer of this product.
-Ben