Hi Juana,
The second example has two N's in the name because the nitrogen atom has two substituents bonded to it that aren't included in the parent name (two methyl groups).
The third example has only one N in the name because the nitrogen atom has only one substituent bonded to it that isn't included in the parent name (one isopropyl group).
-Ben


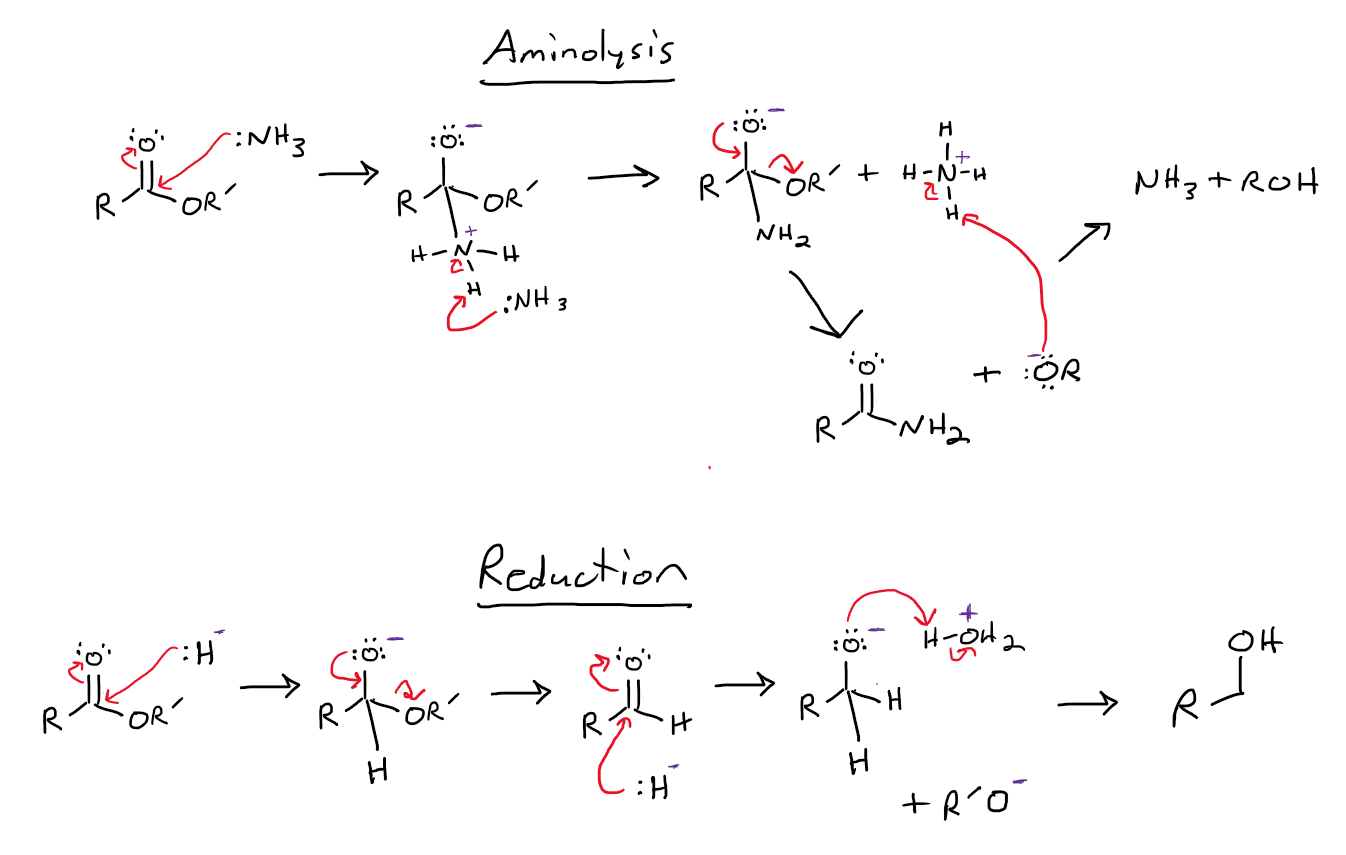
Replied on Lesson: Preparation of Carboxylic Acid Derivatives
20 Mar 04:05
Hi Helen,
See the attached image. The direct way of preparing an amide from a carboxylic acid involves treating the acid with the amine to form a carboxylate salt which, upon heating, undergoes dehydration to yield the amide.
-Ben