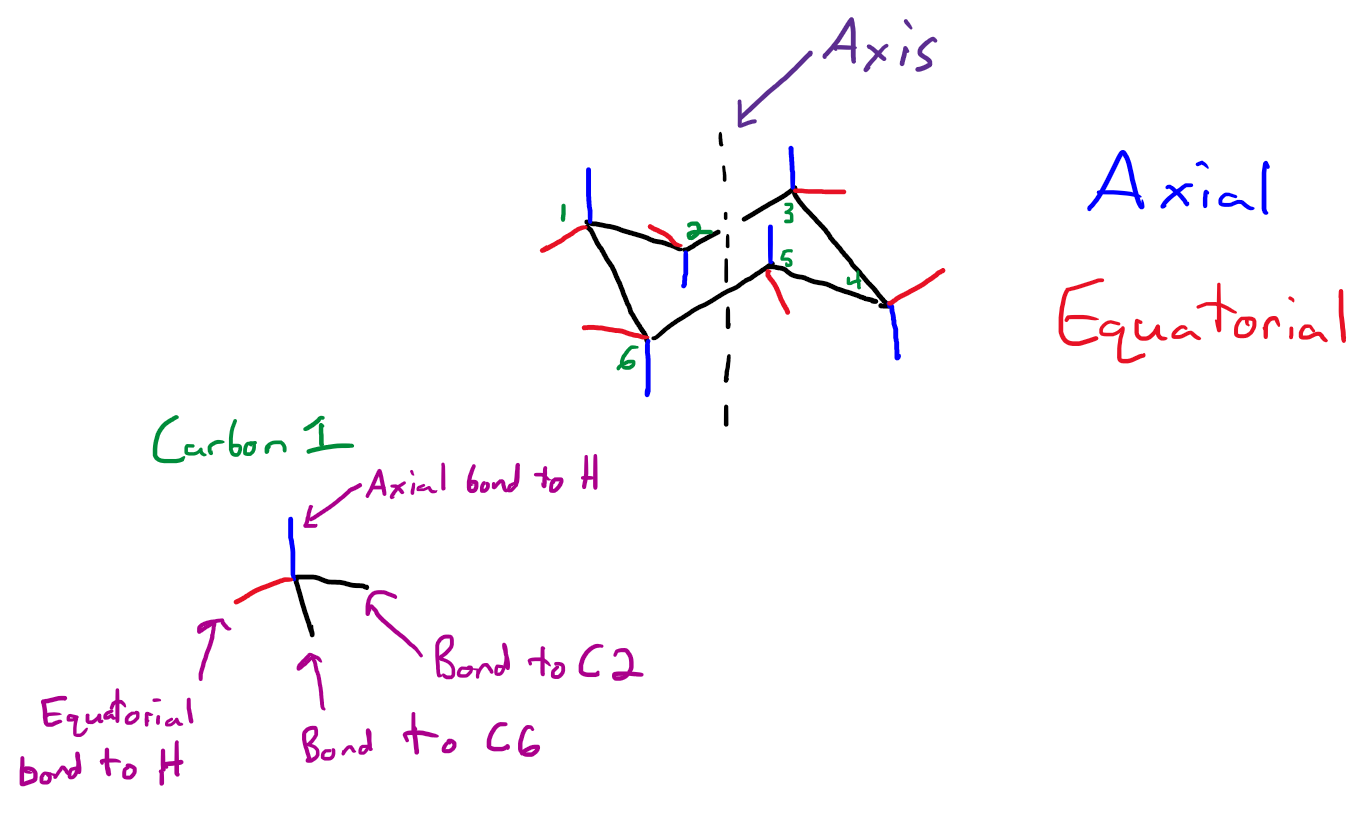
Hi Michelle.See the attached image.
Axial means the bond points parallel to the ring's axis.
Equatorial means the bond lies roughly in the plane of the ring, forming an "equator".
In a cyclohexane chair, each carbon atom has tetrahedral geometry with a bond angle of ~109.5°. In order to keep this geometry, each carbon has two bonds to another carbon atom, an axial bond to a hydrogen atom, and an equatorial bond to a hydrogen atom.
-Ben


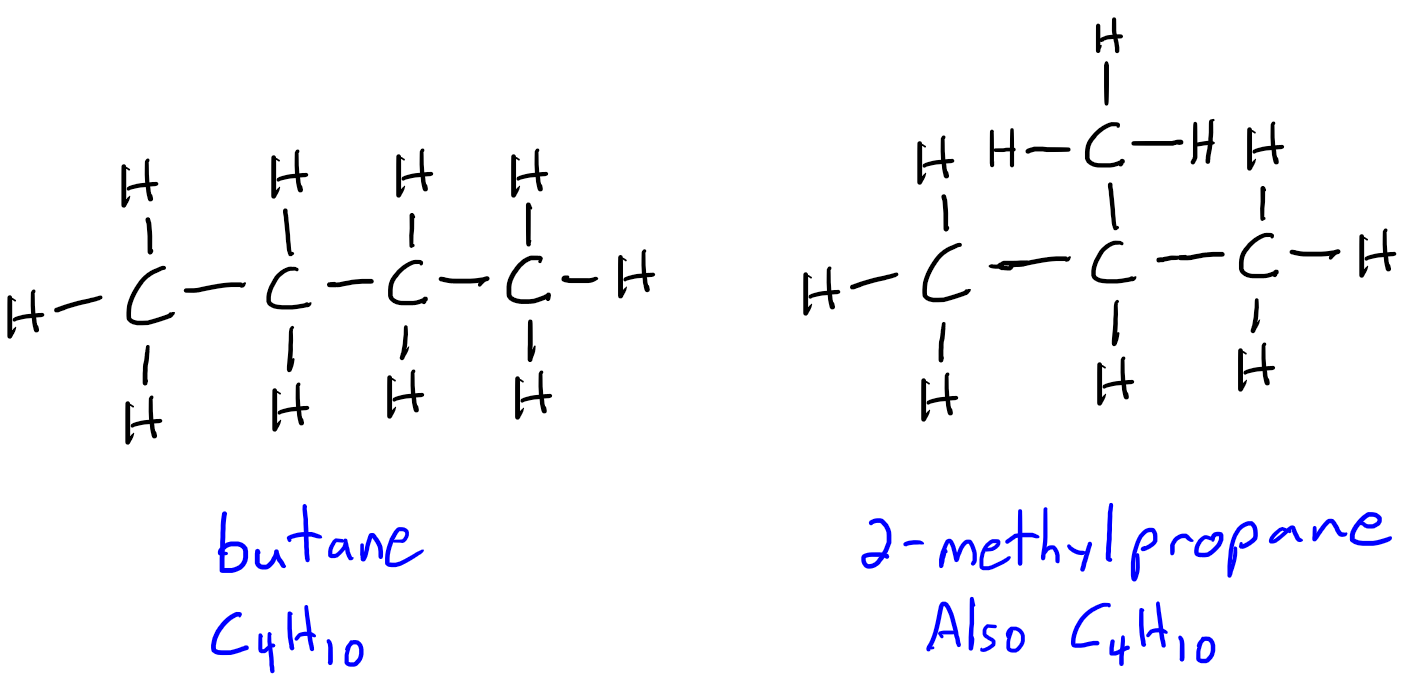
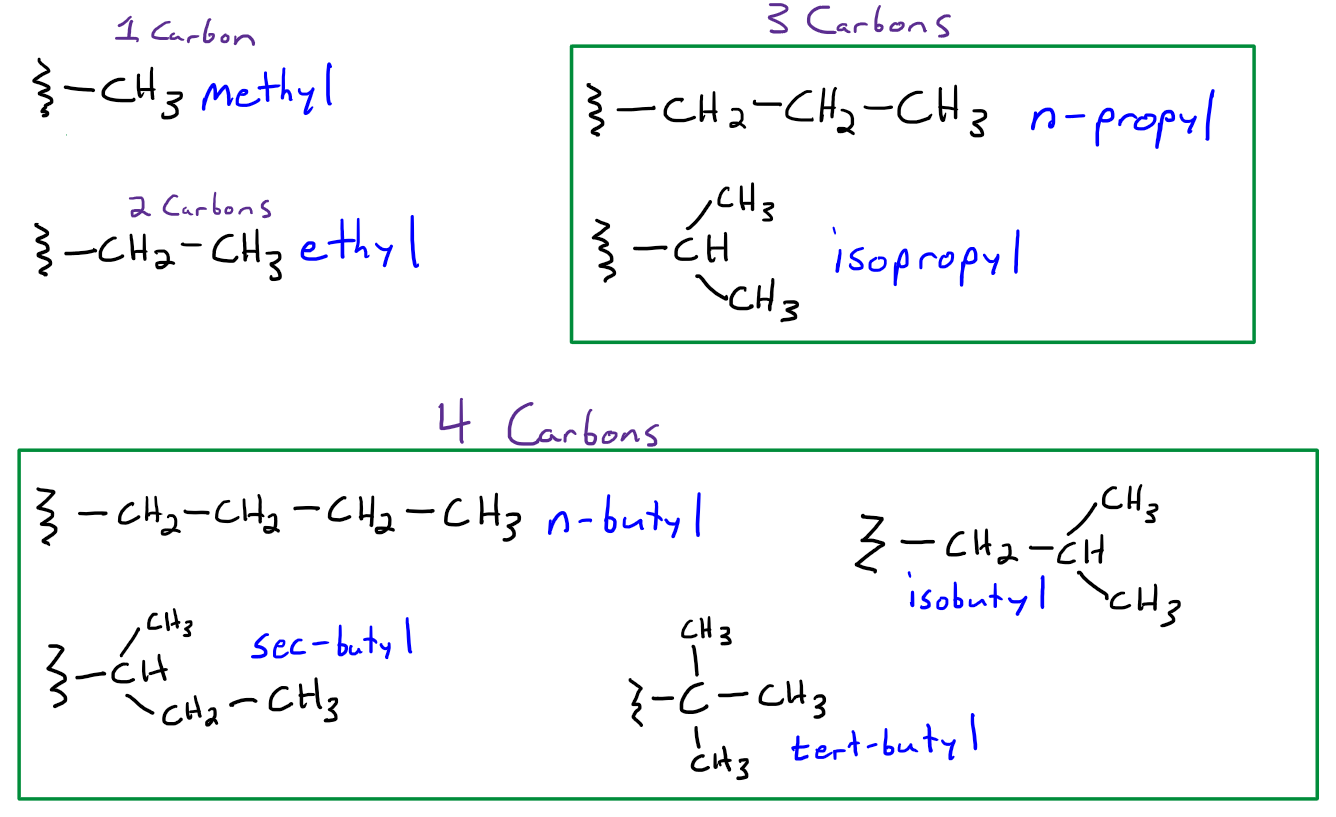
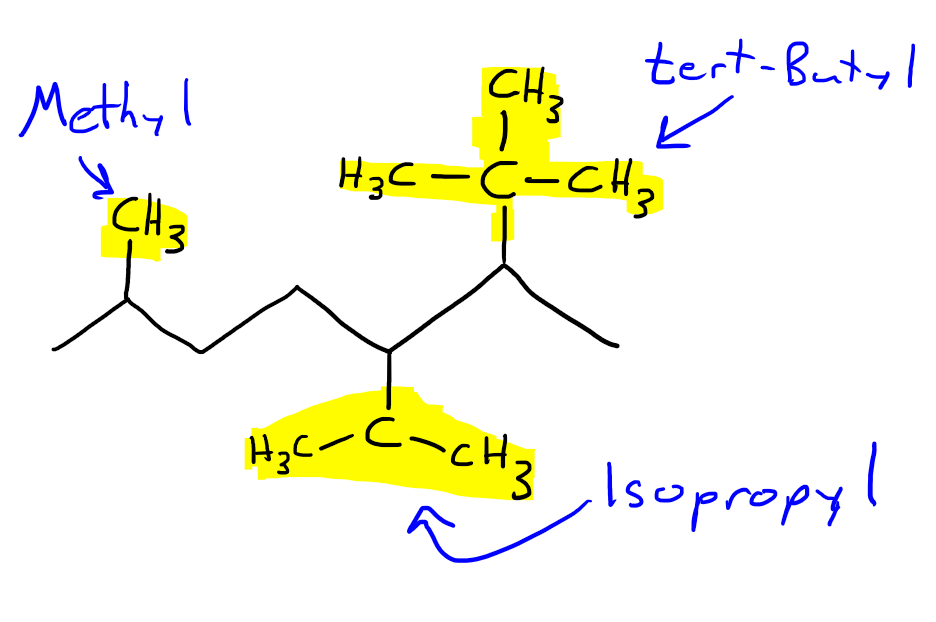

Replied on Study With Us: Identifying Unknown Compounds Using IR and NMR Spectroscopy and Mass Spectrometry | OCHEM
18 Feb 04:53
Hi Mahi,
I'm not sure what you mean by "sp3 peak", but the carbonyl group of an ester typically absorbs at 1735 cm-1 for saturated esters and 1720 cm-1 for aromatic esters, which is somewhat higher than the 1715 cm-1 absorption we typically see for saturated ketones.
Additionally, esters have two C-O bonds that appear as strong peaks in the 1300 - 1000 cm-1 range. For reference, I've attached the IR spectrum of ethyl acetate, a common ester.
While it may be possible that it's an ester from the IR spectrum alone, the mass spec and NMR confirm otherwise.
-Ben