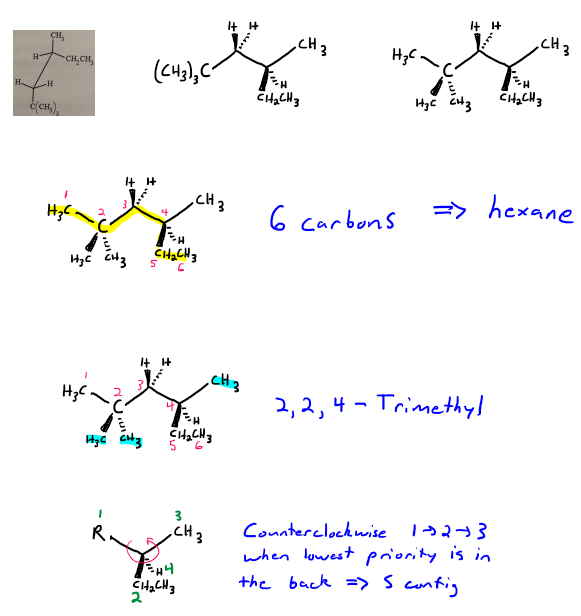
For this one, we can turn the molecule sideways, keeping track of where are substituents are to convert to a structure that uses wedged and dashed lines. This way, we can see the atoms a little bit better.
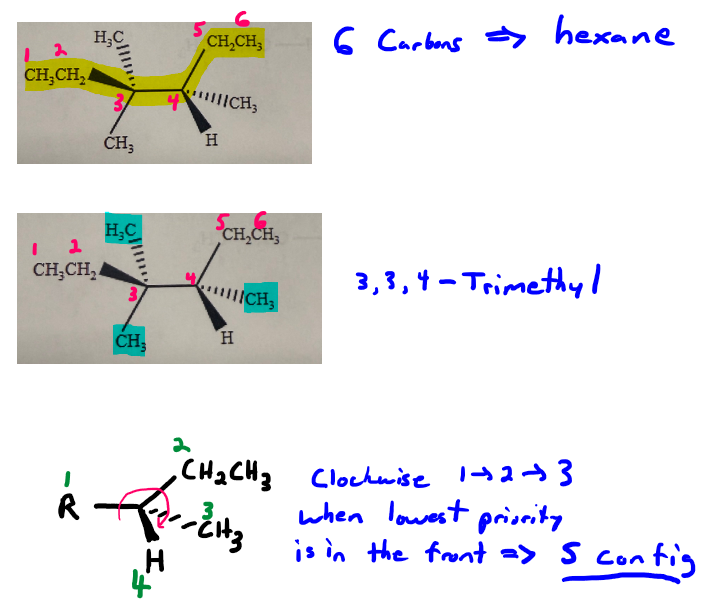
The parent chain is six carbons long, so we have have some sort of hexane. Numbering from the left gives us our first branch point.
There are three methyl groups-two on carbon 2, and one on carbon 4, so it's 2,2,4-trimethyl.
Carbon 4 is a chiral center. Cahn-Ingold-Prelog priorities are as follows: carbon 3 is priority 1, the ethyl group is priority 2, the methyl group is priority 3, and the hydrogen is priority 4. With the lowest priority group in the back, the 1 > 2 > 3 sequence follows the counterclockwise direction, so carbon 4 has the S configuration.
So putting it all together, it's (4S)-2,2,4-trimethylhexane.
-Ben


Replied on Practice Problem: Converting a Molecule Into a Newman Projection
25 Sep 04:25
Hi Paola,
I'm a little confused. Could you show me the original problem you're trying to solve? What is the structure you're starting from and the question itself (i.e. draw a Newman projection viewed along the C2 > C3 bond)?
-Ben