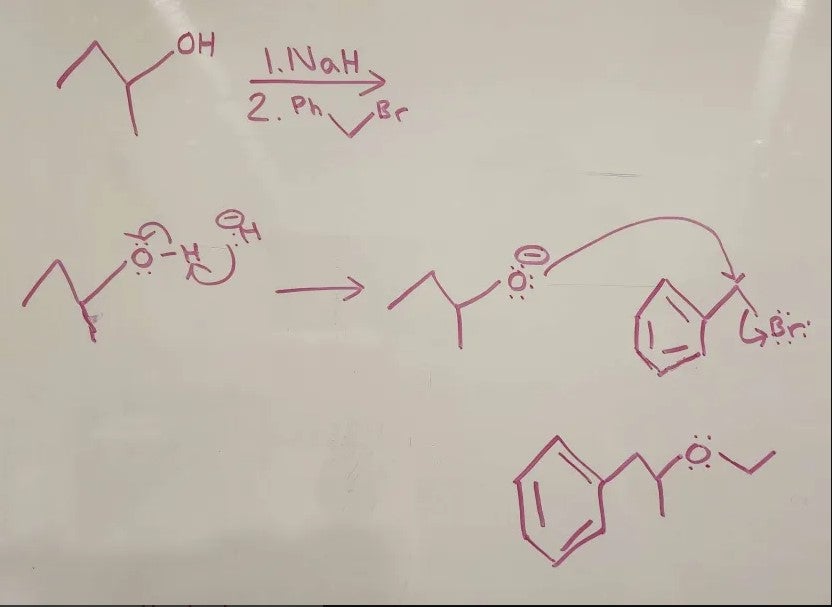
At 21:57, how come we don't attack the secondary because the rule with working for acids, isnt it that you attacked the most substituted? My professor said this
Acid = (most substituted)
1st step. Proton Transfer
2nd step. Sn2
Base = (least substituted)
1st step. Sn2
2nd step. Proton Transfer

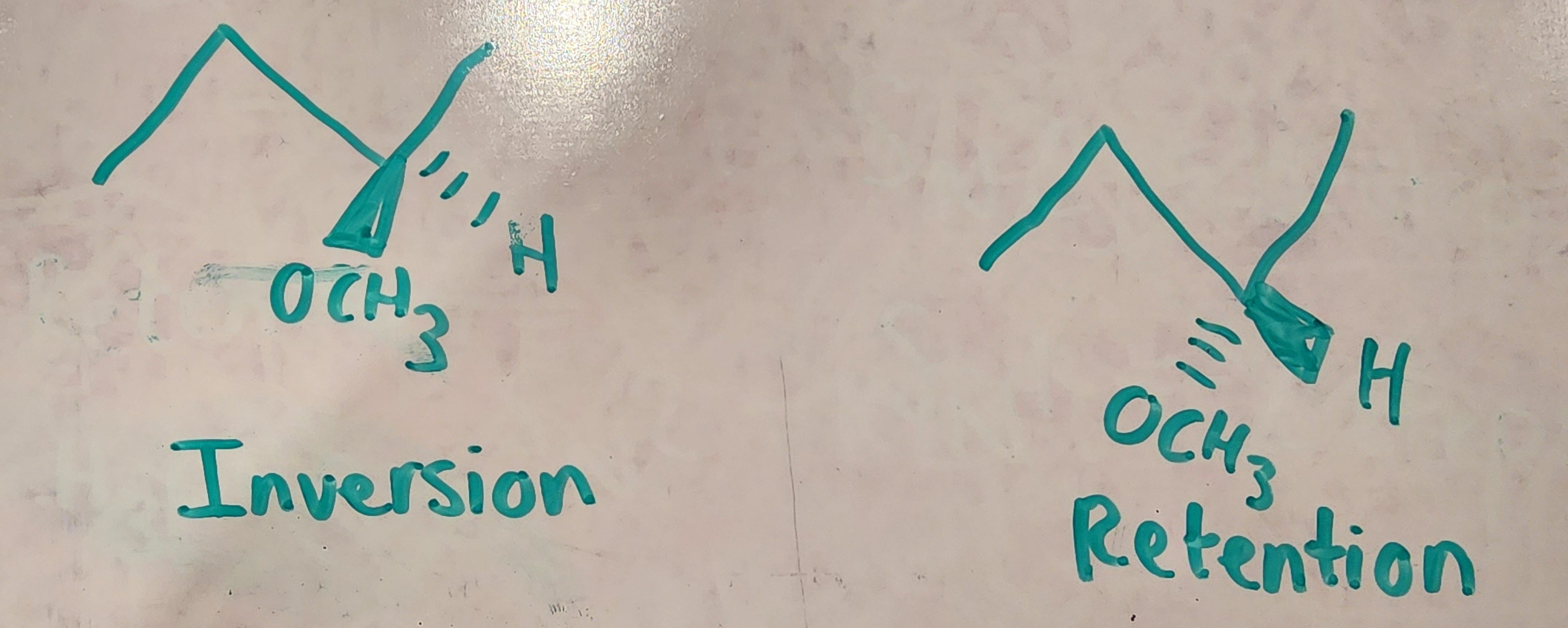
Commented on Lesson: The Formation of Cyclopropanes Using Carbenes and Diazomethane
10 Apr 22:30
This is from my lecture notes. Is this video based on the same thing? Unless there's a video on cyclopropanation. I wasn't able to find your guy's video on it