Hi Michelle,
We know where to add the hydrogens because carbons 2 and 3 are the only ones that have room to accept new hydrogens. Prior to the reduction, all other carbons have four bonds, so they can't form a new bond to a hydrogen atom. We can think of the reduction of an alkyne to an alkane as a two-step process, in which the triple bond converts to a double bond, gaining two new hydrogens, then the double bond coverts to a single bond, gaining two new hydrogens.
-Ben


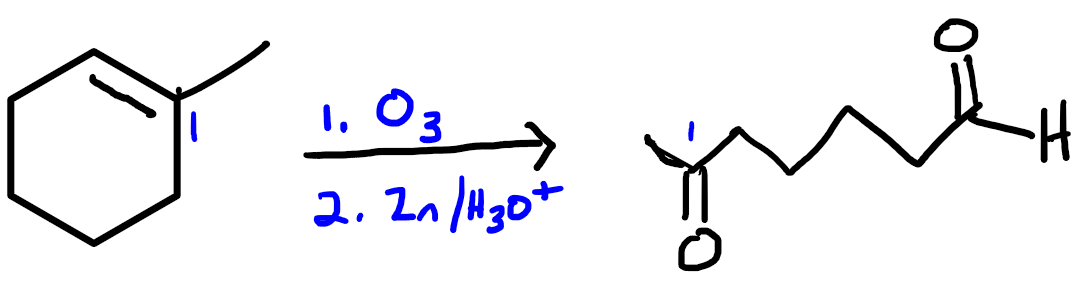
Replied on Lesson: Newman Projections
29 May 04:09
Hi Hope,
The term "gauche" means that the two atoms or functional groups we're looking at are roughly 60° apart when viewing the comformation of the molecule as a Newman projection. It could be any two atoms or groups, as long as one of them is bonded to the front carbon, and the other is bonded to the back carbon.
In the attached image, I've redrawn the isopropyl group as CH(CH3)2 to better illustrate how bulky it is compared to a methyl group.
In the comformation on the left, the methyl group of the front carbon is anti to the isopropyl group and gauche to the methyl group of the back carbon.
In the comformation on the right, the methyl roup of the front carbon is anti to the methyl group of the back carbon and gauche to the isopropyl group.
The comformation on the left is energeitcally favored because the bulky alkyl substituents have a greater degree of separation from one another, causing less steric strain.
-Ben