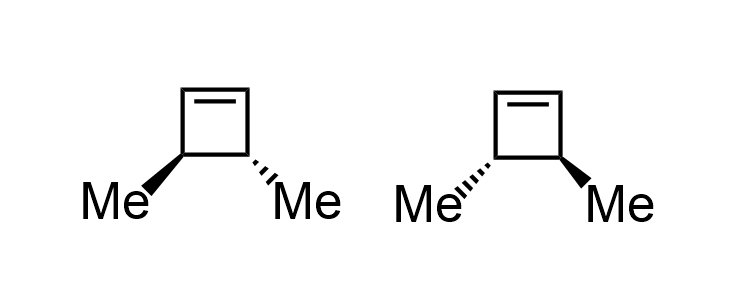
For the heat mediated cyclization at 46:15, you could also form the enantiomer of the compound drawn. Below are both of the possible products.
Replied on Lesson: Electrocyclic Reactions
Replied on Lesson: Wolff-Kishner Reduction
Replied on Lesson: Wolff-Kishner Reduction
Replied on Lesson: Wolff-Kishner Reduction
Replied on Lesson: Naming Compounds With Chiral Centers
Replied on Lesson: R and S Configurations
Replied on Lesson: Reactions of Epoxides
Replied on Practice Problem: Which of the Following Is the Strongest Base?
29 May 17:09
Hi, the easiest way to determine hybridization is to count the number of groups bonded to the atom. Double and triple bonds count as one group.
1 group = s, 2 groups = sp, 3 groups = sp2, and 4 groups = sp3
https://chemmunity.com/programs/organic-chemistry-bond-length-strength-and-hybridization