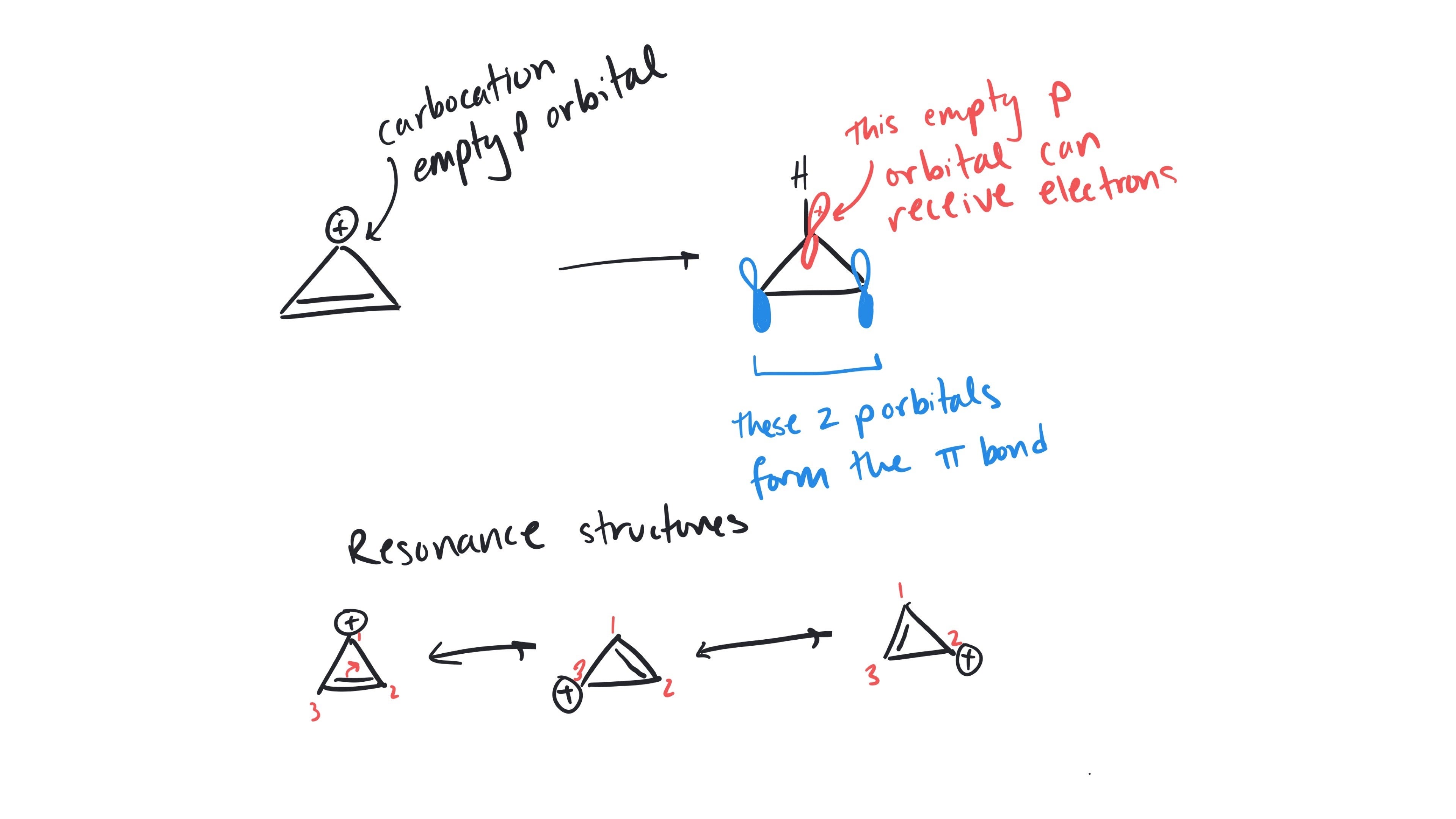
Hi Melanie, this can be confusing for sure! Pi electrons and p orbitals are not the same thing: pi electrons are just electrons in p orbitals. These electrons can go on to make pi bonds with another occupied p orbital. To think about conjugation, you can think about how you might draw a resonance structure. Like drawing resonance structures, no atoms can move but if electrons can move, the system is conjugated.
Melissa is specifically referring to the empty p orbital, which we can also draw as a carbocation. Since carbocations can receive electrons (think resonance!) they are considered conjugated.

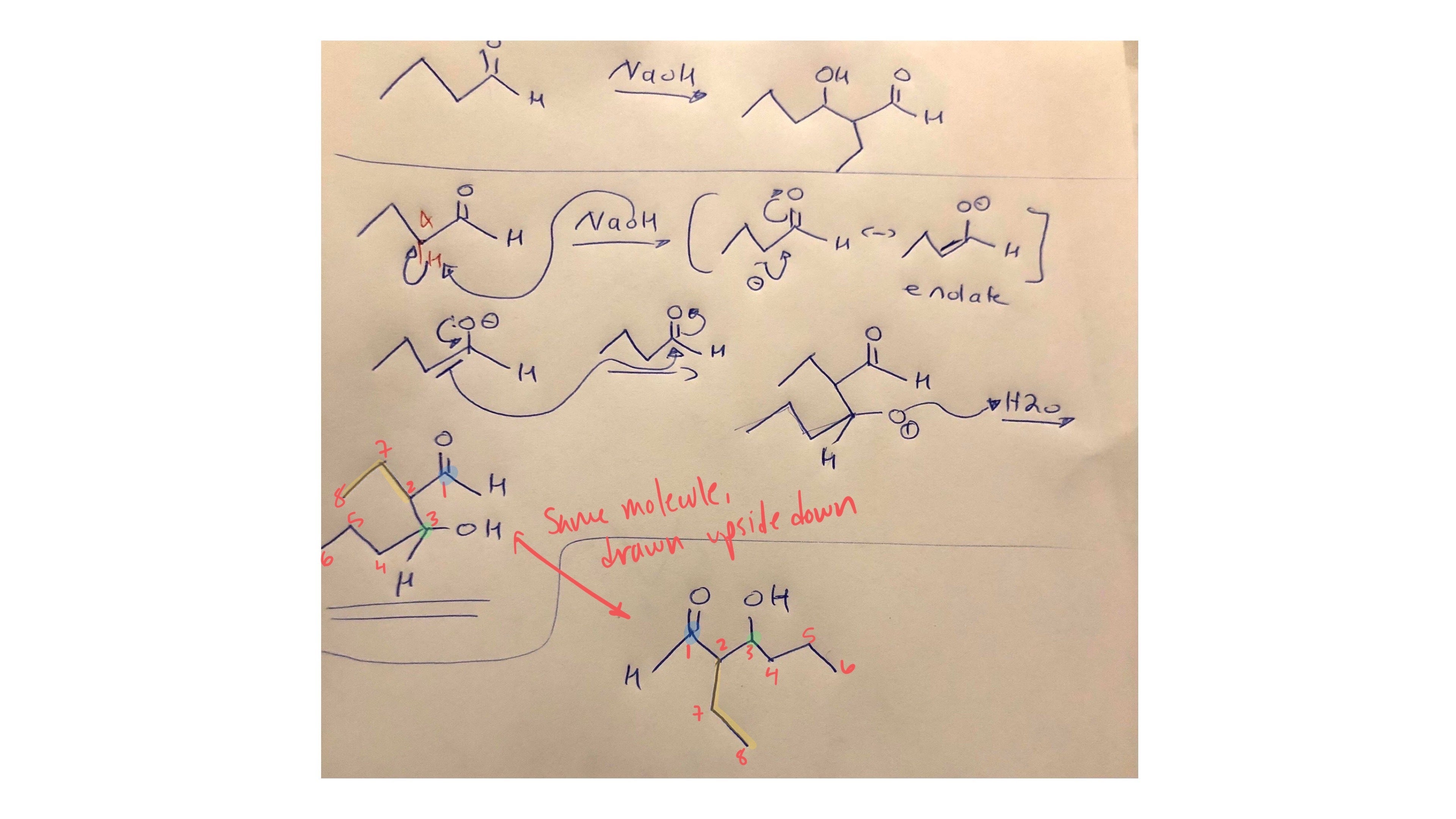
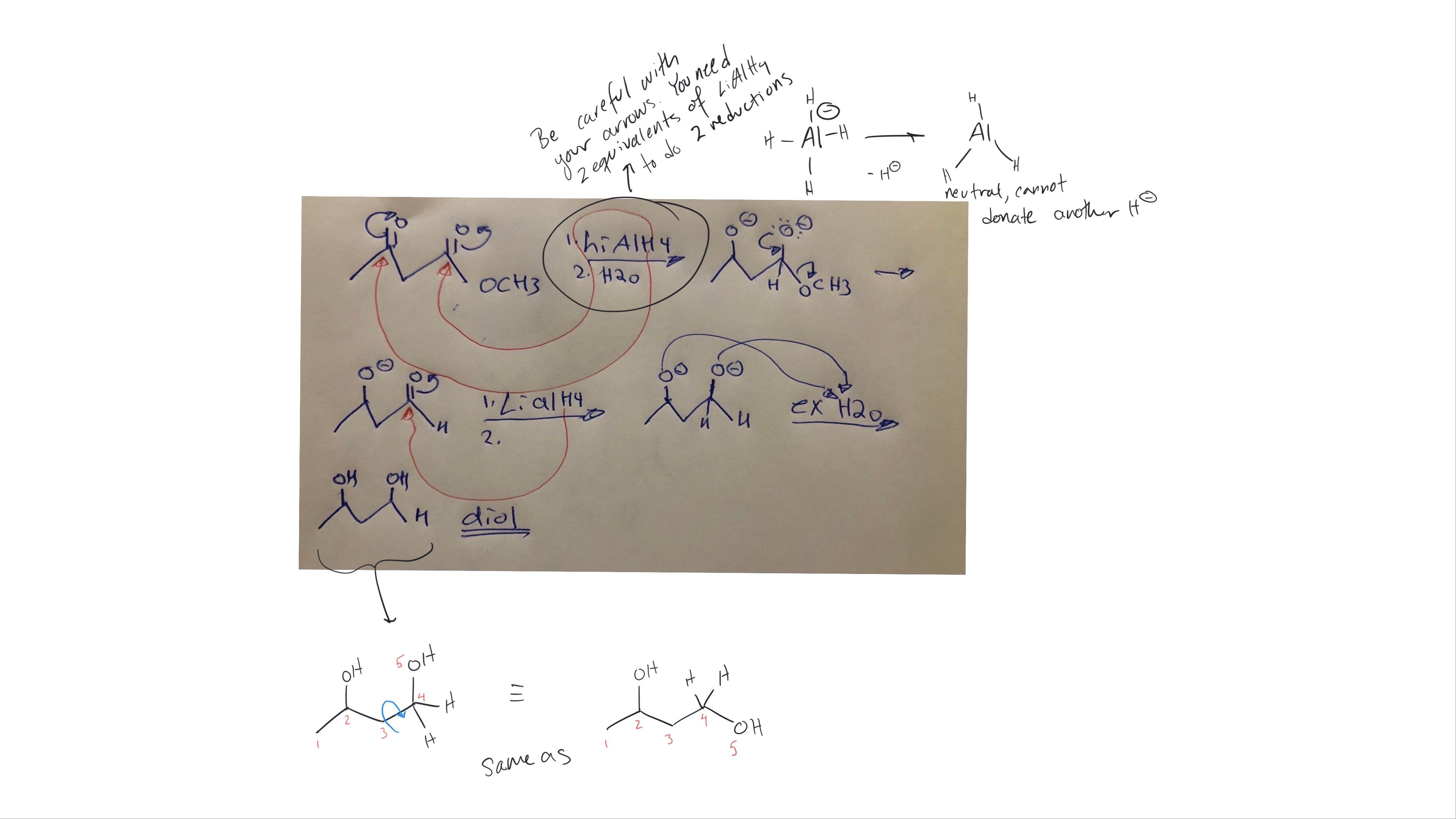
Replied on Lesson: Preparation of Ethers – Williamson Ether Synthesis and Others
11 Mar 20:26
Hi Jag, it looks like you changed the connection between some of your side chains, this answer is not correct. I have drawn the correct structure below and numbered the carbons to show where the new benzyl group is being added to the molecule.
Always draw your structures in the same orientation (view), and number the atoms if you are unsure what has changed, this helps keep track of all the new bonds!