Yosra Abdallatif, thanks for your question. Remember when you're using the clockwise/counterclockwise rule, the lowest priority group has to be in the back (facing away from you). If the lowest priority group is facing towards you, the rule is reversed and counterclockwise is R.

Replied on Lesson: Naming Aldehydes and Ketones
09 Oct 14:47
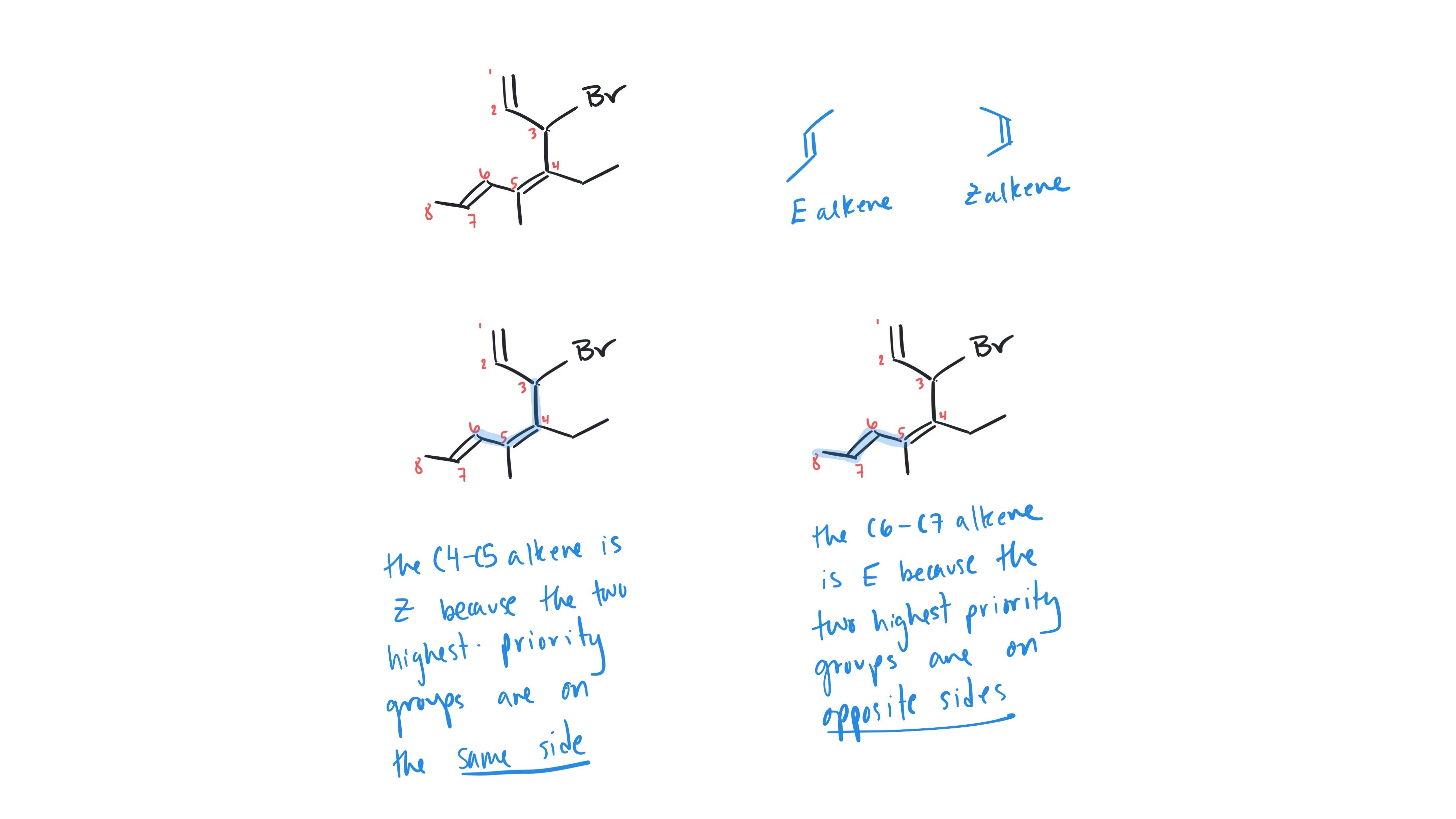
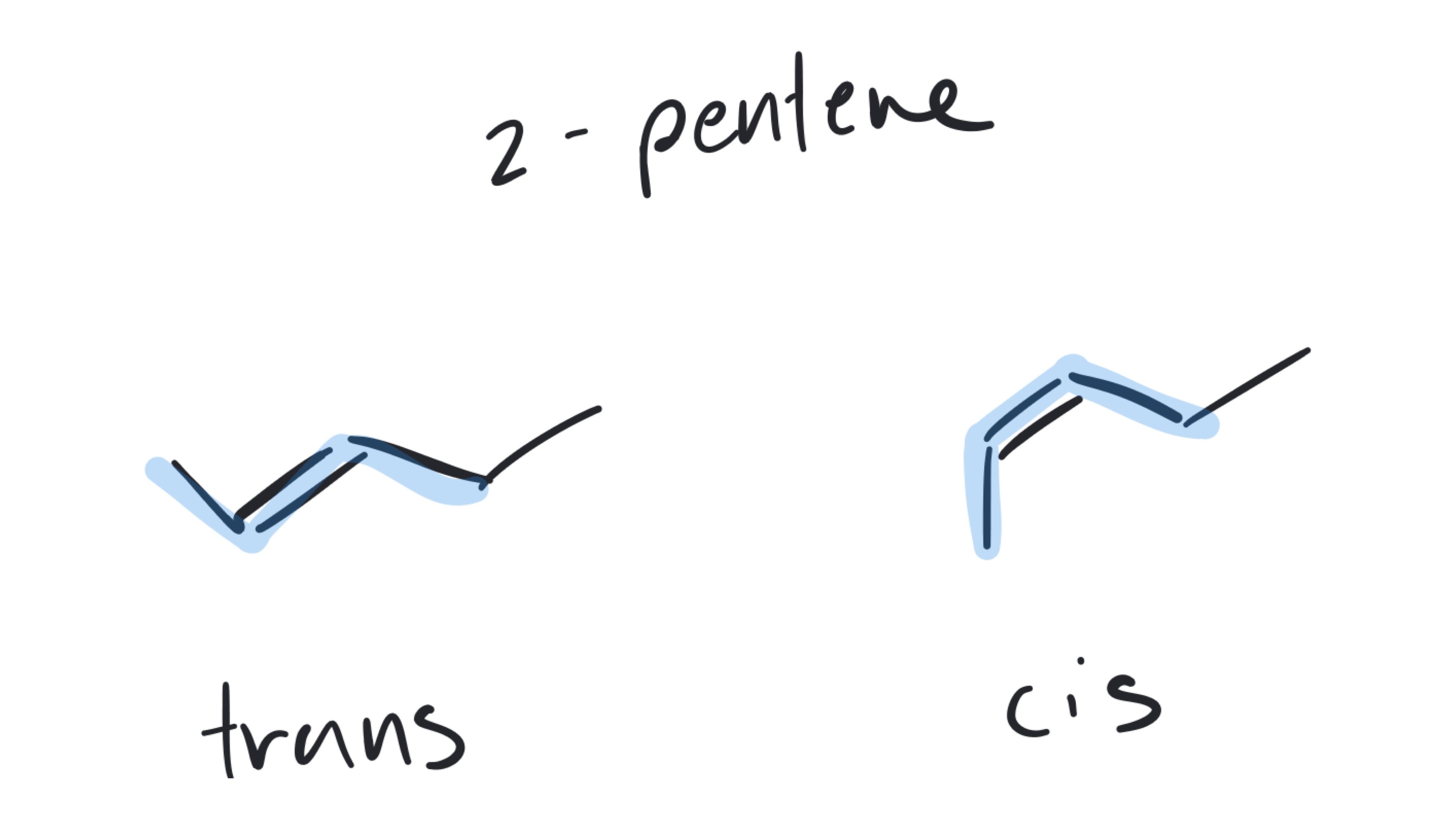
This numbering direction gives the substituents the lowest number possible. If you numbered from the other end of the carbon chain, bromine would be at carbon 3. You are correct, you still want to list the bromine substituent first in the name because of the alphabetical ordering.