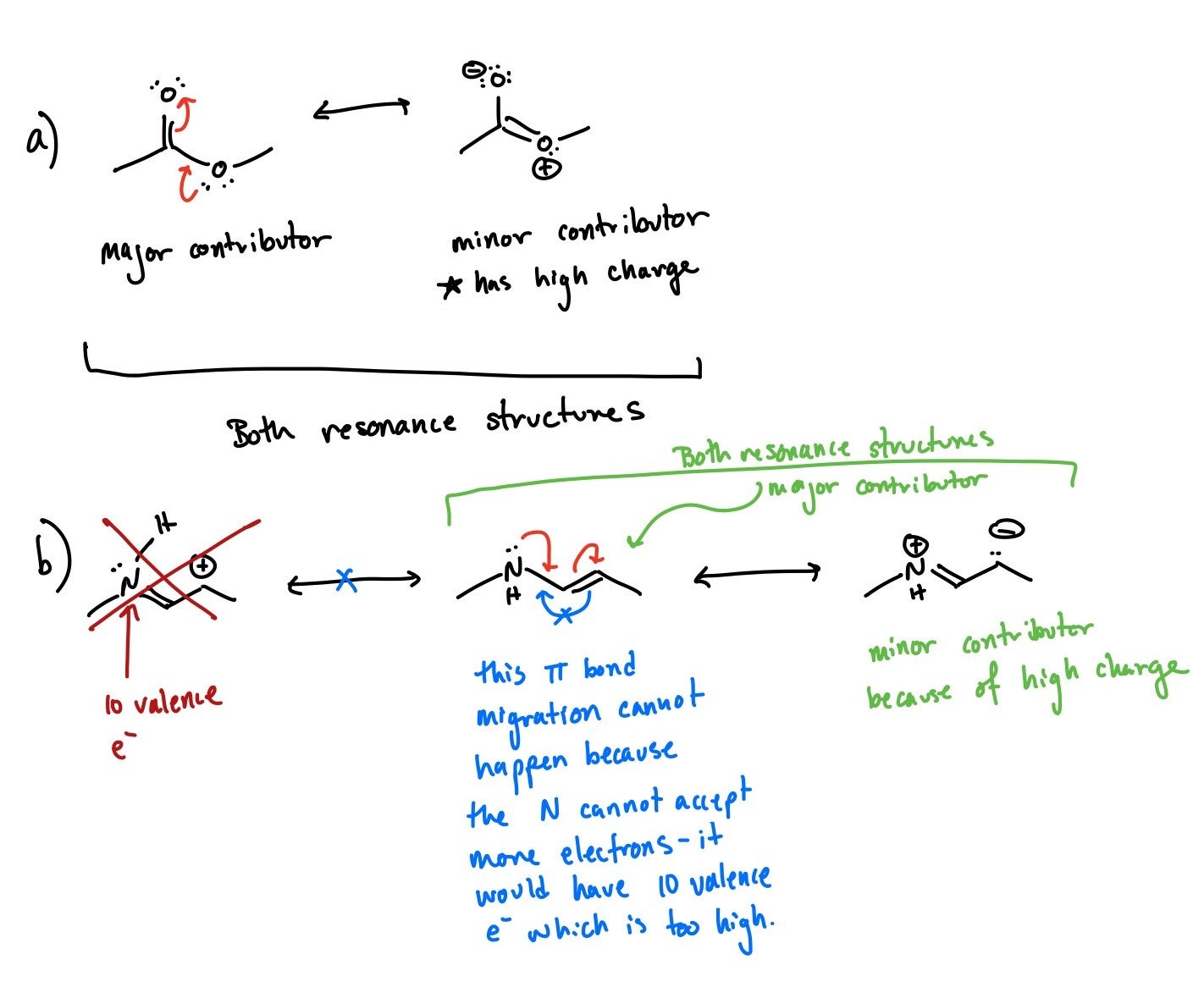
Saliho Toure, thanks for your questions. Remember the question that this video is covering is "what is the major resonance contributor". For A, the lone pairs on the oxygen can participate in resonance, but this resonance structure has a high formal charge, so it is not the major resonance contributor.
For B, there is another resonance structure that is not explicitly drawn in the video, again because it has high formal charge so it cannot be the major resonance form (contributor). If you are asked to draw all the resonance structures, it would be correct to draw the major and all minor resonance contributors.

Replied on Practice Problem: Identifying Enantiomers and Diastereomers
25 Sep 10:00
em , D does not have a diastereomer because of its symmetry. We call this type of molecule 'meso' because you can draw a plane of symmetry through the molecule.