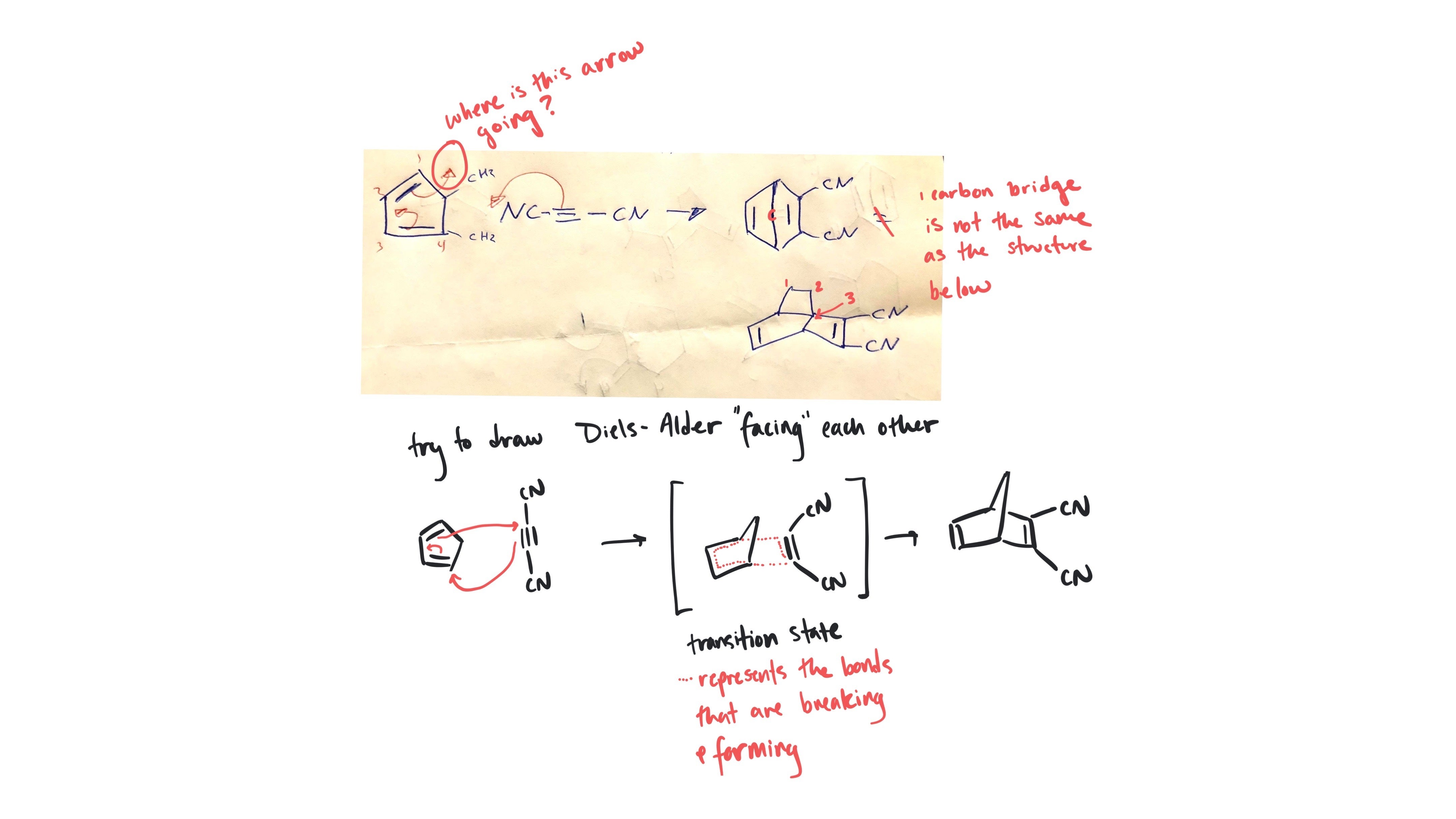
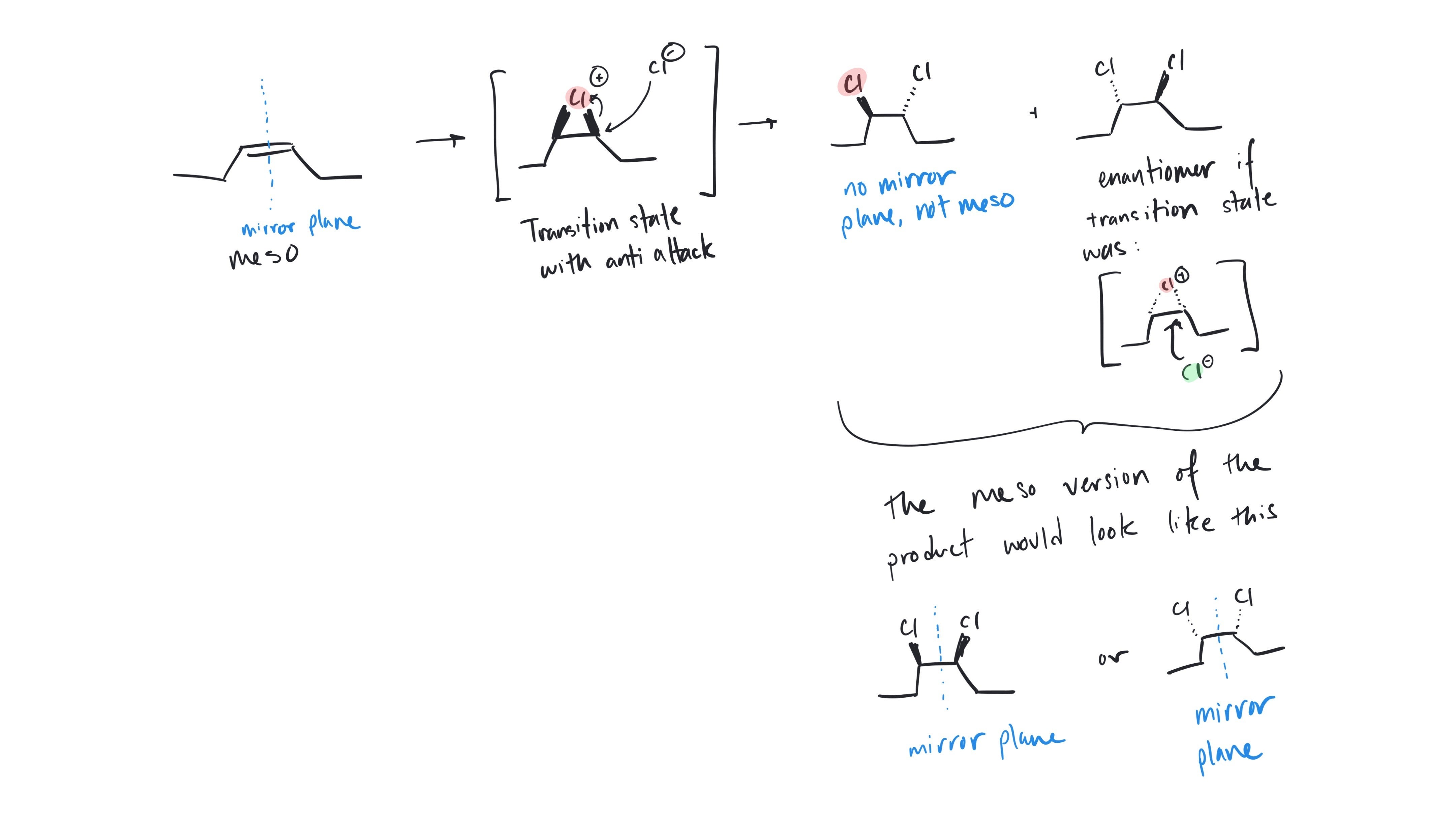
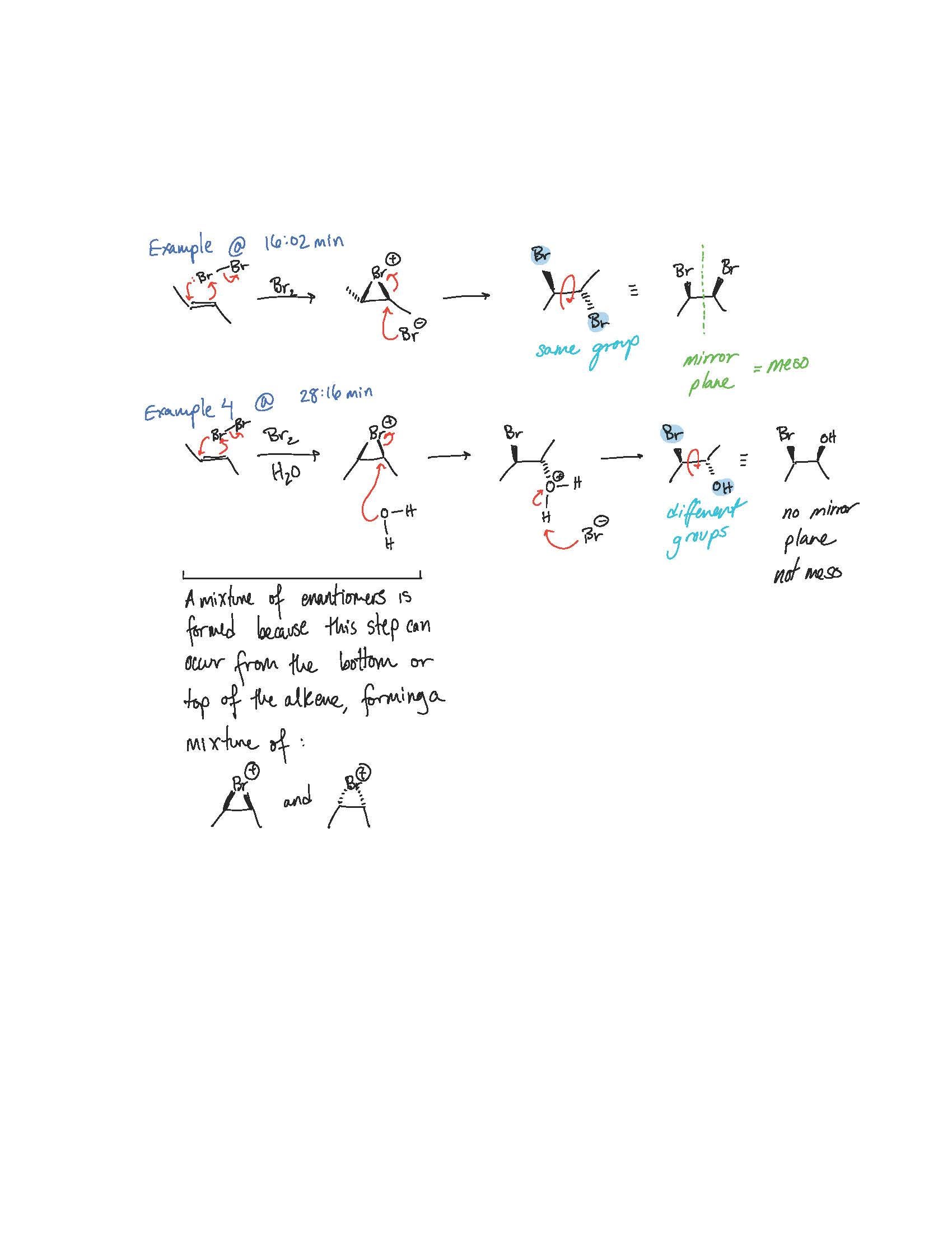
Mariam, I see. You have drawn a completely different starting material, the cyclic and open-chain are not the same molecule and can't interconvert. Your arrows look correct for the starting material you have drawn, but see my notes below on the correct product.

Replied on Lesson: Nucleophilic Aromatic Substitution
23 Jan 20:19
Hi Mona, thanks for your question. Electrophilic aromatic substitution (EAS) and nucleophilic aromatic substitution (NAS or SNAr) have different mechanisms, and therefore different trends when it comes to reactivity.
If you are asked to rank the reactivity of substituted benzene molecules, look at the other reagents for clues. Remember that EAS needs to have a strong electrophile (a group that receives electrons, or gets attacked, sometimes these are positively charged). In EAS, you are correct, moleucles with electron donating groups (EDG) are the most reactive.
For NAS or SNAr reactions, the other reagents must have a strong nucleophile (a group that donates electrons or attacks another molecule, sometimes these are negatively charged or have lone pairs of electrons). In SNAr, electron withdrawing groups (EWG) are the most reactive because they make the ring a better electrophile.