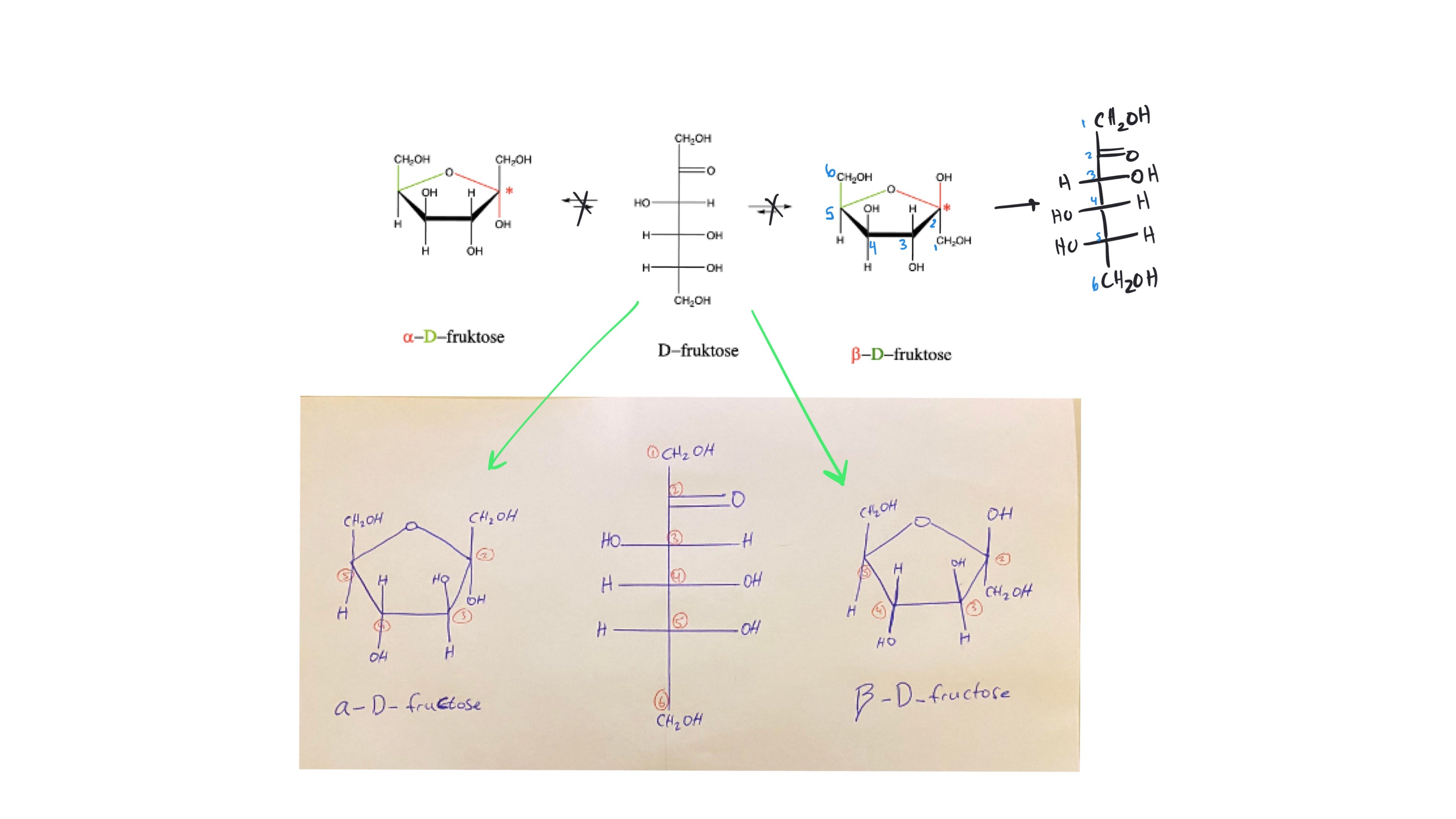
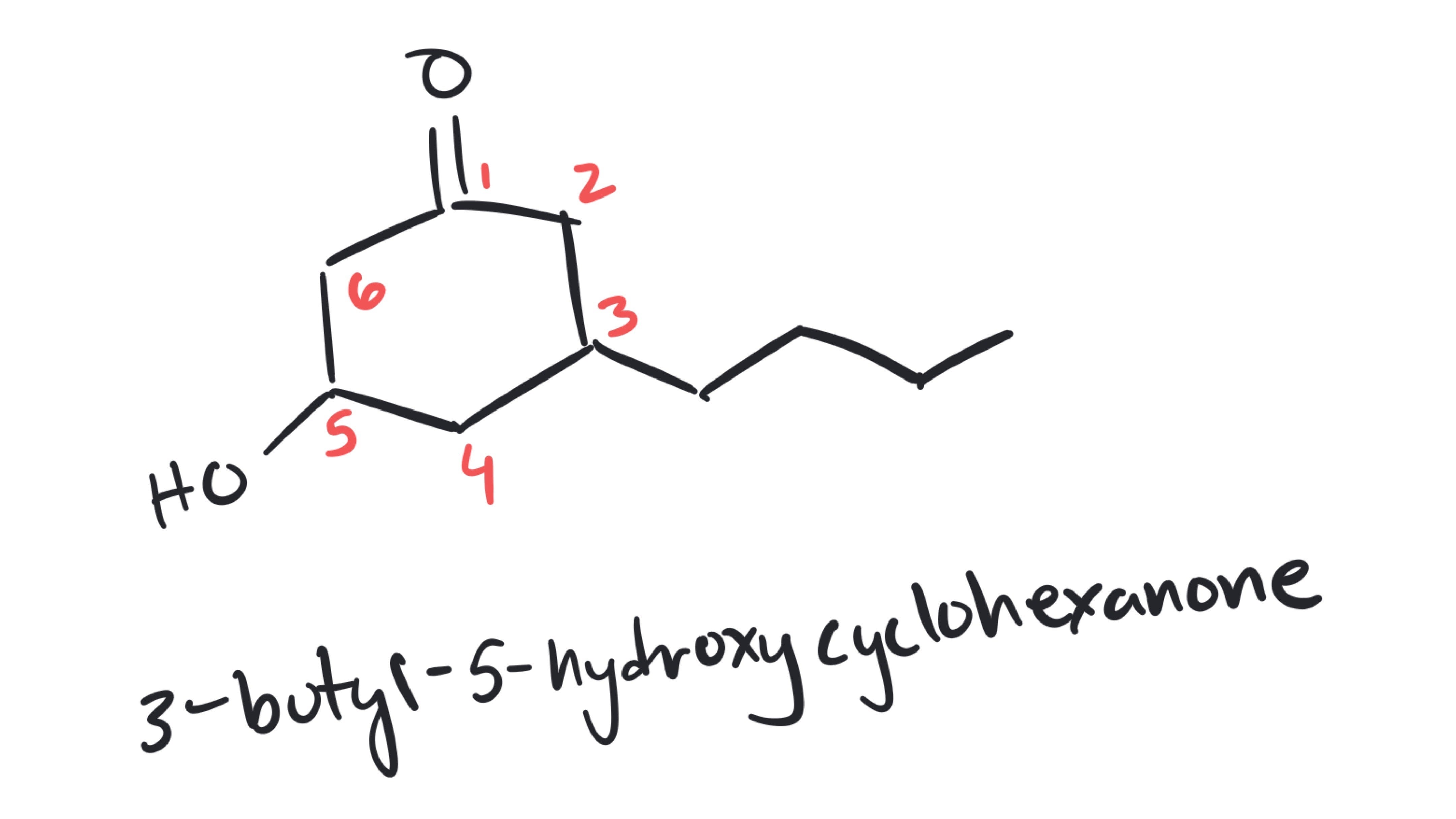
Hi Johnathan, the name you've written out with the 'di' prefix is absolutely correct. To be even more precise, you can include the position of the alcohol by naming the compound 6-chloro-2,2-dimethylcyclohexan-1-ol (note, if the alcohol has no defined position, it is assumed to be at carbon 1). Thanks for asking for clarification!

Replied on Lesson: SN1 Reactions
25 Oct 11:57
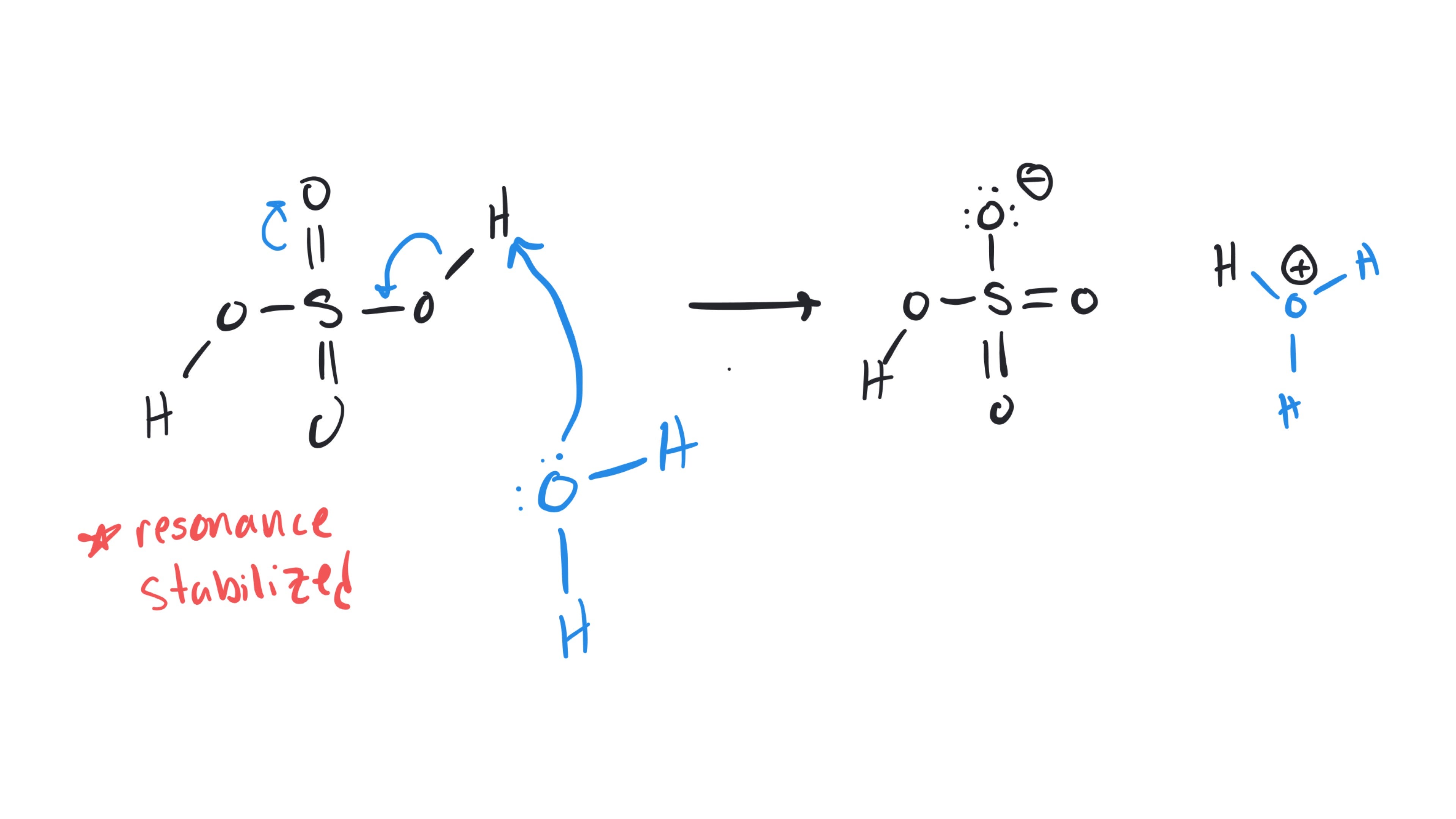
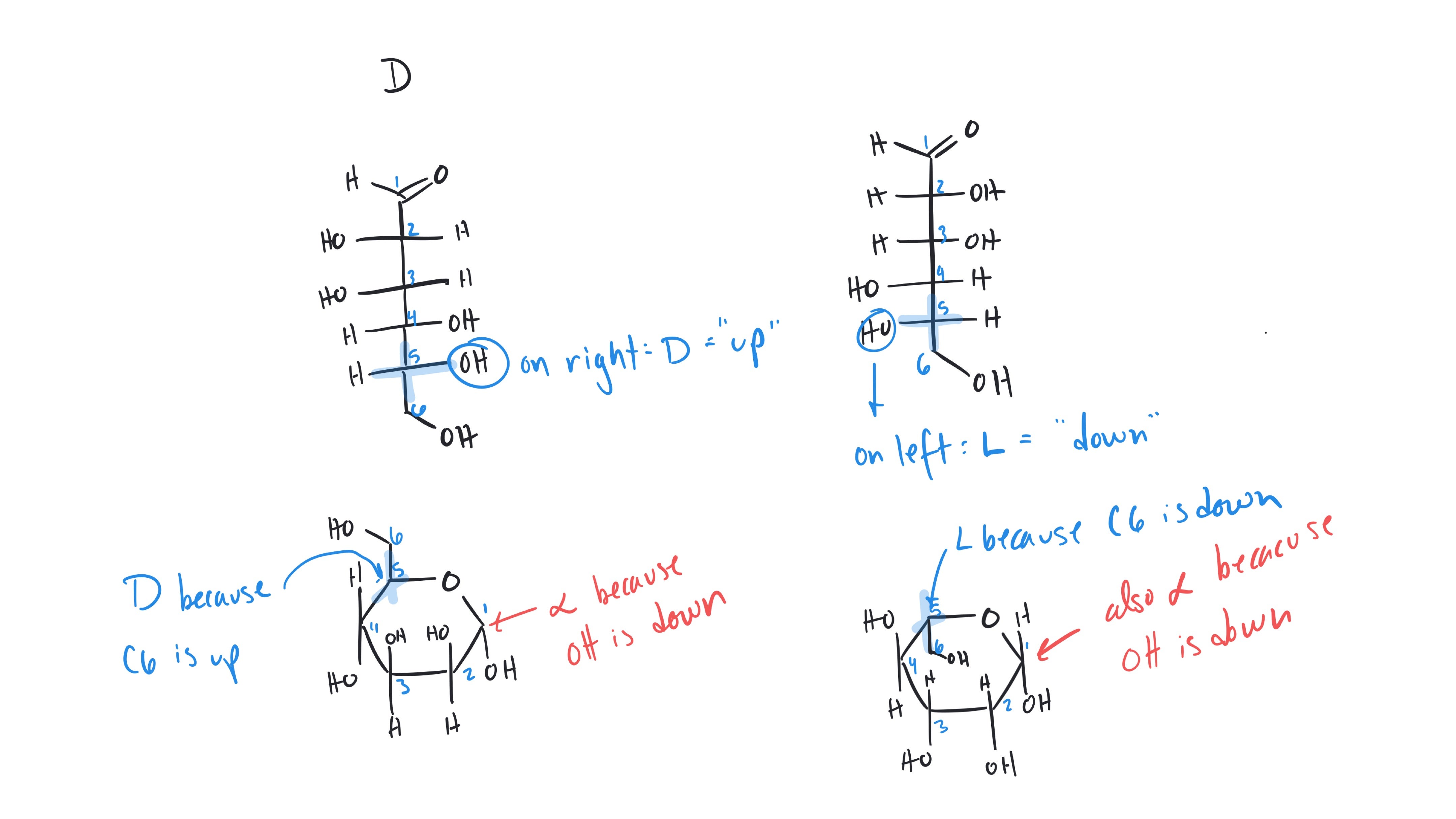
Yosra Abdallatif, yes exactly! The carbocation still has three bonds, 2 bonds to carbon and 1 bond to hydrogen which is not shown in the 'skeletal form' as a convention. Below I've drawn out all the 'implied' hydrogens on the molecule and highlighted the hydrogen that migrates in this problem.